Home > Ocean Acidification
Ocean Acidification
Ocean acidification threatens the entire Southeast Alaskan marine ecosystem – an ecosystem that we depend on both for subsistence and livelihoods. SEATOR members have recognized the importance of reacting proactively to ocean acidification to reduce impacts to traditional food sources. We will be collecting continuous data on several ocean parameters and using that data to write a comprehensive ocean acidification adaptation plan. All of our data will be publicly available on the Alaska Ocean Observing System database and will be shared with the Alaska Ocean Acidification Network.
What is ocean acidification?
Ocean acidification, or “OA” is the name given to the lowering of the ocean’s pH due to the increase in atmospheric carbon dioxide (CO2) since the industrial revolution. The pH scale measures the acidity of a liquid and spans from 0 to 14. Liquids with a pH of zero are extremely acidic, while those with a pH of 14 are extremely basic Freshwater is neutral is has a pH of 7, while lemon juice has a pH of about 2, and lye’s pH is 13. The pH scale is logarithmic, so a change from a pH of 8 to a pH of 7 means the solution has gotten ten times more acidic.
The ocean is slightly basic, with a natural pH of about 8.2. As carbon dioxide dissolves in water, though, it forms carbonic acid, lowering the pH of the ocean. While the ocean is unlikely to become truly “acidic”, shell-making organisms require high pHs to make their shells. As OA progresses, shellfish, plankton, and even finfish are starting to show signs of stress. The pH of the ocean has changed from approximately 8.2 to 8.1 since the start of the industrial revolution, representing a 26% increase in acidity and a rate of change more than 100x faster than anything the Earth has seen for at least tens of millions of years. If CO2 emissions remain unchecked, the Intergovernmental Panel on Climate Change (IPCC) predicts that the ocean could become 150% more acidic over the next century.
Why are we at risk?
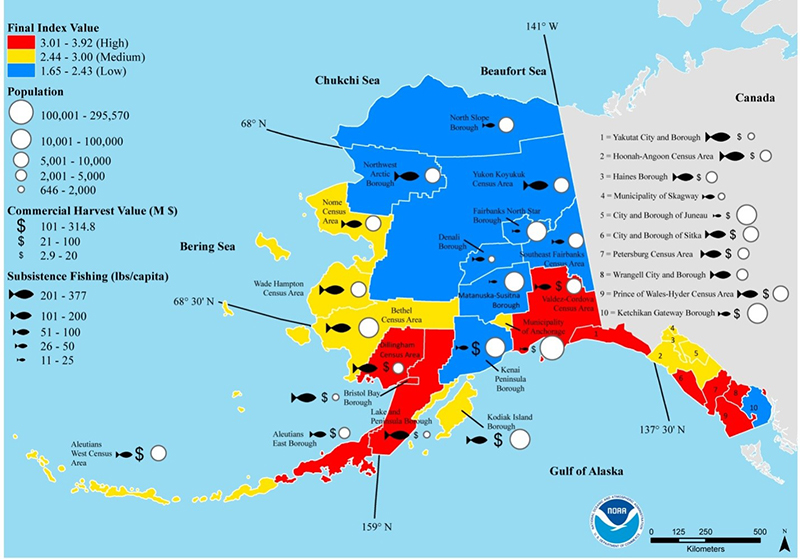 Alaska as a whole is especially vulnerable to OA due to our shallow seas, cold water (which can hold more CO2), high rates of primary production, large amount of glacial melt. A 2015 NOAA study on Alaska’s regional vulnerability to climate change and ocean acidification cited Southwest and Southeast Alaska as regions at the highest overall risk for negative economic and social effects from ocean acidification, as illustrated by the map at right. The study noted both regions’ high dependence on healthy fisheries and lack of viable alternative employment opportunities. The study also noted each region’s high dependence on subsistence resources, most of which are predicted to be negatively impacted by OA.
Alaska as a whole is especially vulnerable to OA due to our shallow seas, cold water (which can hold more CO2), high rates of primary production, large amount of glacial melt. A 2015 NOAA study on Alaska’s regional vulnerability to climate change and ocean acidification cited Southwest and Southeast Alaska as regions at the highest overall risk for negative economic and social effects from ocean acidification, as illustrated by the map at right. The study noted both regions’ high dependence on healthy fisheries and lack of viable alternative employment opportunities. The study also noted each region’s high dependence on subsistence resources, most of which are predicted to be negatively impacted by OA.
In a nutshell, Alaska depends on its fish and fisheries, and Southeast Alaska depends on those resources more than most. OA is predicted to both decimate the small, shelled plankton that form the base of our marine food web and to directly limit growth rates and navigational ability in finfish. All shellfish species are also highly dependent on ocean pH and are likely to suffer as our waters become more acidic.
What are we doing in response to Ocean Acidification in Alaska?
To monitor current and long-term future OA trends, the Sitka Tribe will collect continuous pH, pCO2, dissolved oxygen, temperature, and salinity data using a moored buoy outside of Sitka Sound. As funding allows, we will steadily add more monitoring equipment to our buoy to further inform our understanding of current ocean conditions. We are working with several partners to help us calibrate our equipment and gather the most accurate data we can, including the SEATT members, NOAA, the University of California Santa Cruz, Alaska Ocean Observing System, Alutiiq Pride Shellfish Hatchery, and the Sitka Sound Science Center.
The short term goal of our ocean observing equipment is to help Tribal leaders develop ocean acidification and climate change adaptation plans for their communities. Only with current, high-quality information can we accurately predict and react to changes in marine traditional resources.
Collaborators and additional information:
- Alaska Ocean Observing System
- Alaska Ocean Acidification Network
- Prof. Burke Hales, Oregon State University College of Earth, Ocean, and Atmospheric Sciences
- Dr. Wiley Evans, Hakai Institute
- Intergovernmental Panel on Climate Change
- Alutiiq Pride Shellfish Hatchery
- UCSC
- Sitka Sound Science Center
- BIA Climate Change Program
